Our research program uses (1) inorganic chemistry, organic chemistry, and (2) chemical biology to address important metal-mediated processes with energy, biological, and medical relevance. An interdisciplinary, problem-based approach will be employed for the synthesis and characterization of new organic and inorganic compounds, with the ultimate goal to address unsolved problems with broad implications to our society.
After reading through our research overview, take a look at our Publications and take our Virtual Lab Tour. If you have questions about our work, please feel free to Contact Us!
Inorganic & Organometallic Chemistry Research
Novel Catalysts for the Conversion of Natural Gas into Liquid Fuels & Other Oxidative Transformations
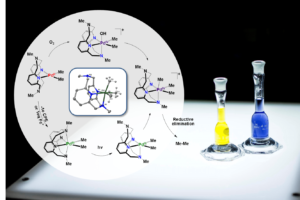
The inorganic/organometallic research projects in our group focus on the development of transition metal complexes with unique reactivity by employing judiciously designed multidentate ligands that control the electronic properties and reactivity of metal ions in various oxidation states. We are combining strategies from biology for the activation of small molecules (e.g., O2 and CO2) with successful approaches from organometallic chemistry for the functionalization of unactivated organic molecules (e.g., CH4 and other hydrocarbons), with the ultimate goal of employing such catalysts in renewable energy applications such as the conversion methane other light alkanes into liquid fuels (Figure 1a), which would allow for a more efficient use of natural and shale gas reserves as an inexpensive energy resource, resulting in a major impact on our society and the environment.
During the past several years we have synthesized a series of uncommon high-valent PdIII and PdIV complexes, and we have investigated in detail their organometallic reactivity. For example, we have shown unprecedented aerobic oxidation of PdII precursors to generate detectable PdIII and PdIV intermediates that lead to C-C and C-heteroatom bond formation. This aerobically-induced C-C bond formation reactivity supports a catalytic cycle for the aerobic oxidative dimerization of hydrocarbons, and current studies aim to develop novel Pd catalytic systems for aerobic oxidative C-H coupling reactions.

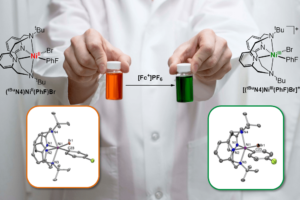
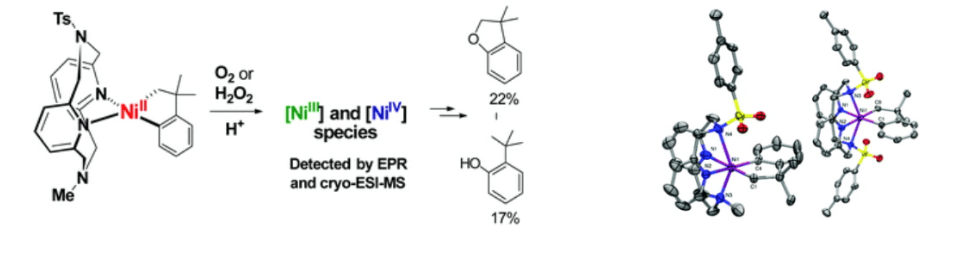
We have also recently reported the isolation, detailed characterization, and reactivity studies of a series of novel organometallic mono-aryl/alkyl and di-alkyl/aryl NiIII and NiIV complexes supported by flexible multidentate ligands. With an appreciable number of isolated organometallic NiIII and NiIV complexes in hand, we are now investigating their involvement in important chemical transformations, as well as uncover new reactions involving these high-valent Ni systems. Specifically, our immediate objectives are to a) probe the role of well-defined high-valent Ni species in stereocontrolled catalytic C-C bond formation reactions, and b) develop aerobic oxidative reactions mediated by high-valent Ni intermediates, with direct relevance to C-H functionalization and C-C/C-heteroatom bond formation reactions (Figure 1b).
Finally, we are synthesizing and characterizing odd-electron organometallic Rh and Ir complexes. Such complexes are exceedingly rare, and the isolation of these species will open up a wide range of organometallic reactivity studies, especially oxidatively-induced catalytic C-H functionalization reactions.
Read more in the Mirica Group’s Publications
Chemical Biology Research
Multi-functional Compounds as Novel Theranostic Agents for Beta-Amyloid Peptide Aggregation in Alzheimer’s Disease.
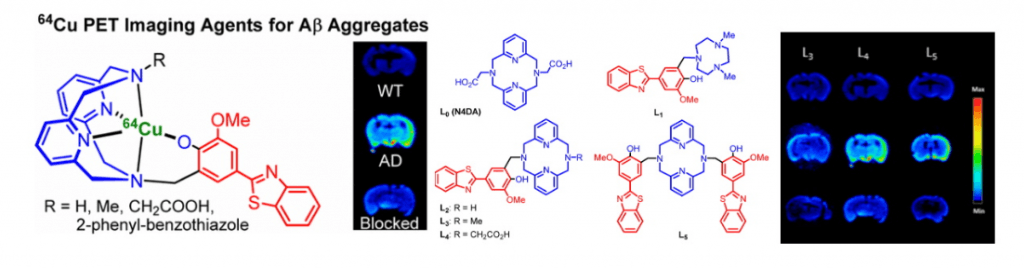
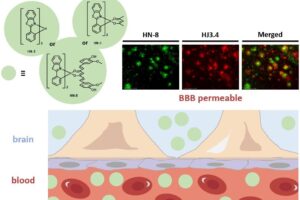
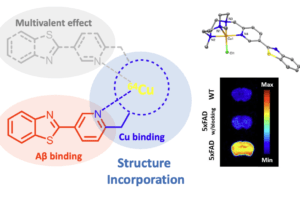
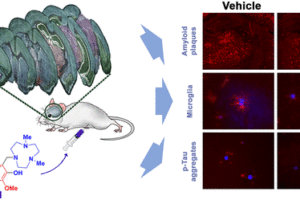
Alzheimer’s disease (AD) is the most common neurodegenerative disease. The brains of AD patients are characterized by the deposition of amyloid plaques that contain the amyloid beta (Ab) peptide. Abnormal interactions of copper (Cu), zinc (Zn) and iron (Fe) ions with the Aβ peptide are proposed to play an important role in the pathogenesis of AD, and disruption of these metal-peptide interactions holds considerable promise as a therapeutic strategy. The long-term goal of this project is to develop novel therapeutic and diagnostic agents for AD based on bifunctional compounds that interact with Ab species and also bind transition metal ions (Figure 2). We have recently reported bifunctional compounds that have high affinity for both Aβ aggregates and metal ions, and have studied their effect on the neurotoxicity of the Aβ aggregates. Second-generation bifunctional compounds show a limited ability to disaggregate Aβ fibrils and exhibit no cellular toxicity, and these compounds show promising initial results in in vivo Aβ clearance studies.

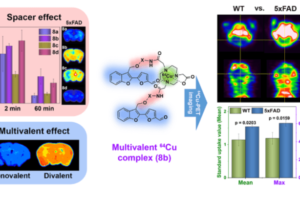
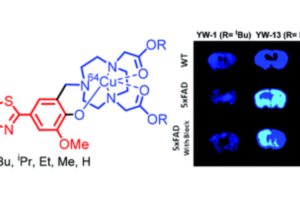
We are also interested in developing metal-containing compounds for positron emission tomography (PET) imaging of Aβ aggregates – including soluble Aβ oligomers, as an early diagnostic tool for AD. For example, bifunctional compounds that exhibit very high affinity for metal ions could be radiolabeled with the 64Cu radionuclide – a longer-lived ideal PET tracer, to generate an amyloid-binding imaging agent (Figure 2). Importantly, the targeted proposed compounds could be employed as both therapeutic and diagnostic PET imaging agents, since the potential therapeutic agent can be 64Cu-radiolabeled and used as an imaging agent to monitor the in vivo response to therapy. In this regard, we have recently synthesized a series of Ab-binding bifunctional compounds that strongly chelate Cu ions. 64Cu radiolabeling and biodistribution studies reveal the ability of these complexes to cross the blood-brain barrier, while initial animal PET imaging studies have shown that these 64Cu-labeled bifunctional compounds show the specificity necessary to image Aβ plaques in vivo. We also particularly interested in bifunctional compounds that will specifically target soluble Aβ oligomers for both therapeutic and imaging applications, a strategy that has not been employed to date.
Read more in the Mirica Group’s Publications